Resources
To search for clinical trials and detailed information on products in development, visit our Prevention Research & Development Database

Results
showing 1-10 of 327
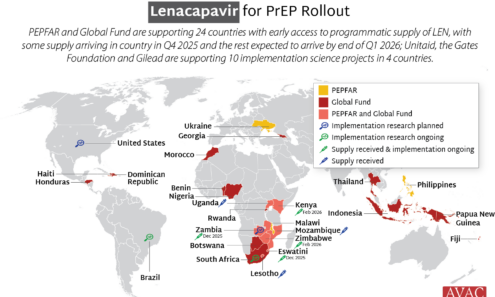
Source of Lenacapavir for PrEP Supply to Early Adopter Countries
The Global Fund, with support from CIFF, and PEPFAR have jointly committed to reaching up to two million people with injectable lenacapavir for PrEP over three years. Supply of LEN began arriving in countries in late 2025 with service delivery planned to start in early 2026.
Prevention Option:
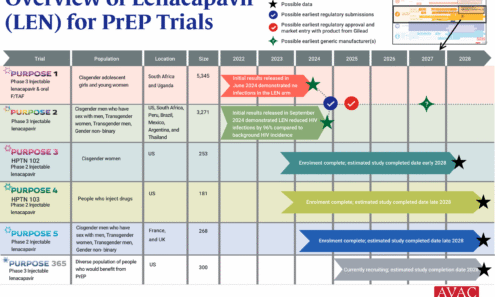
An Overview of Lenacapavir for PrEP Trials
The PURPOSE trials evaluate the safety and efficacy of injectable lenacapavir (LEN), an investigational antiretroviral (ARV) drug being studied as a potential PrEP product. This graphic shows the latest status of all five trials including the groundbreaking results of PURPOSE 1 and PURPOSE 2.
Prevention Option:
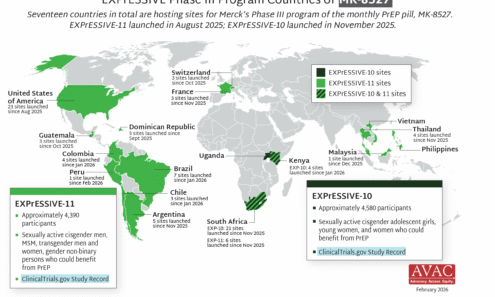
EXPrESSIVE Phase III Program Countries of MK-8527
Seventeen countries are hosting sites for the Phase 3 efficacy trials of a monthly PrEP pill, MK-8527, being developed by Merck (also known as MSD outside of the US and Canada). Merck announced the launch of the Phase 3 trials at IAS 2025 in Kigali. MK-8527 was found to be safe and well-tolerated in Phase 2 clinical trials.
Prevention Option:
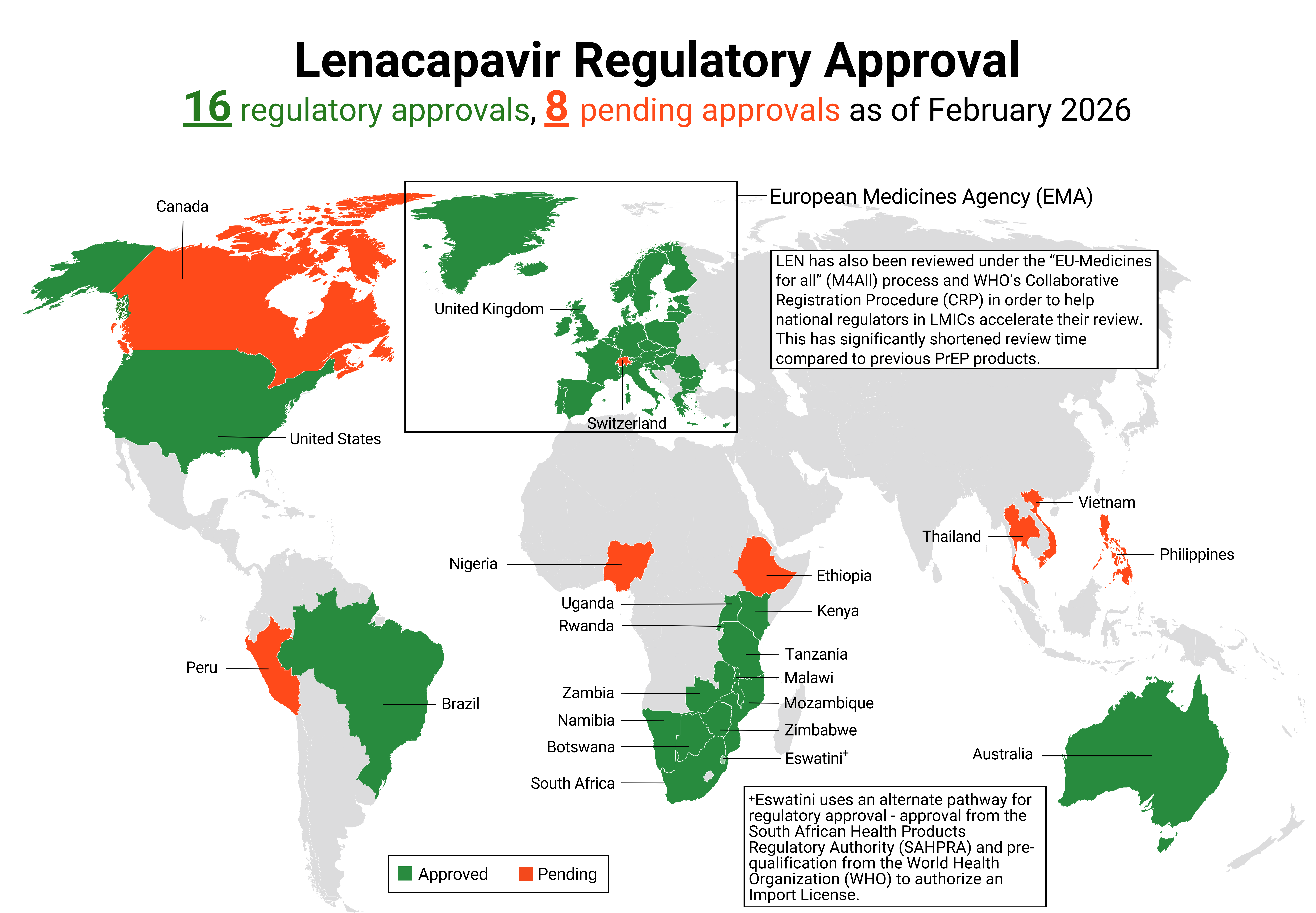
Lenacapavir Regulatory Approval
Regulatory approvals, pending decisions, and appeals as of February 2026.
Prevention Option:
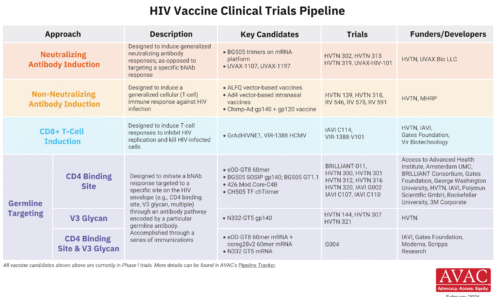
HIV Vaccine Clinical Trials Pipeline
This graphic summarizes the state of HIV vaccine research, detailing the different immunological approaches in clinical trials, the specific candidates being studied, and the collaborative networks of funders and developers working toward an effective vaccine.
Prevention Option:
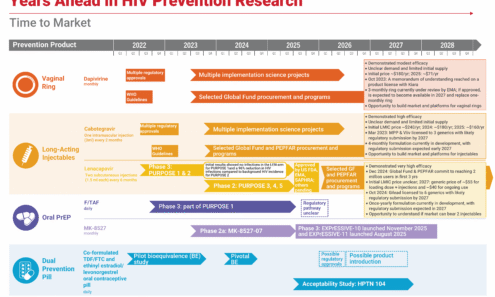
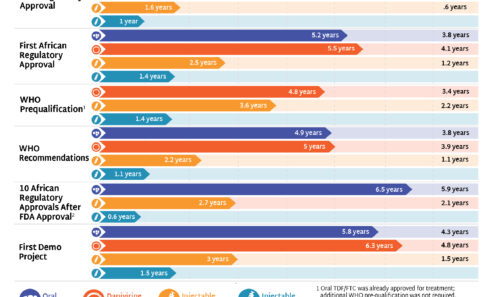
Years Ahead in HIV Prevention Research: Time to Market
This timeline shows the potential time points when the next-generation of HIV prevention options might find their way into new programs.
Prevention Option:
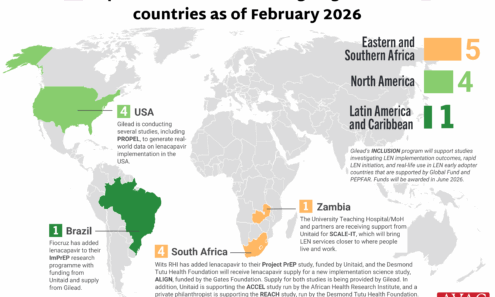
Lenacapavir Implementation Studies
Ongoing and planned implementation studies for the lenacapavir as of February 2026.
Prevention Option:
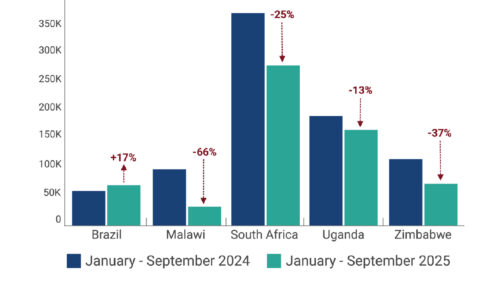
Change in PrEP Initiations from 2024 to 2025 in Five Countries
Between the period January-September 2024 to January-September 2025, PrEP initiations fell between 13% and 66% in selected high-volume PrEP countries where ministries of health (MoH) were able to provide data. Among the five countries depicted, four saw significant decline in PrEP uptake. All four relied on PEPFAR PrEP programs and were disrupted by stop work orders (SWO).
Prevention Option:
Speeding Up Access to PrEP
The timeline for hitting key milestones in product introduction is moving faster for injectable LEN than for any previous PrEP products, starting from the announcement of efficacy results in Phase III trials. LEN’s accelerated timeline compared to oral PrEP, DVR, and CAB reflects a field-wide effort to learn lessons from previous PrEP rollout and not repeat the mistakes of the past.
Prevention Option:
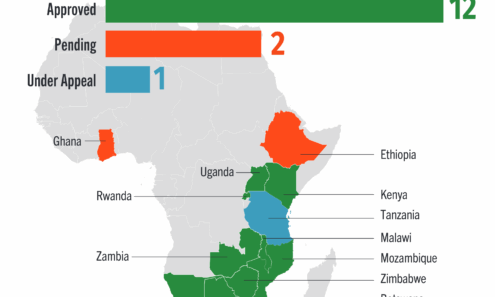
Dapivirine Vaginal Ring Regulatory Approval
Regulatory approvals, pending decisions, and appeals as of January 2026.
Prevention Option:
showing 1-10 of 327